Making Beer
/Recipes Our Recipes
/Labels Making labels for your homebrew
/Ingredients Ingredients on hand
Homebrew-Discuss Our mailing list for brewing related things.
/BrewChecklist and /BottleChecklist: handy checklists to keep brewing on track
What's Brewing
Recently Bottled
- 2014-03-30: Joey's Jasmine-Oolong Pale Ale
Fermenting
- 7.5 gal carboy: nil
Better Bottle BT0: nil
Better Bottle SK0: ClintonEbadi's spring pale ale
Better Bottle SK1: nil
Better Bottle UL0: nil
Better Bottle UL1: nil
Better Bottle SF0: nil
- Small Better Bottle:
- 5 gal carboy:
1 gal jug UL0: RachelBrown's Ancient Orange Mead (??-??-??)
1 gal jug UL1: KristenPeavler's Lavender Rose mead (2012-03-17)
1 gal jug UL2: nil
1 gal jug UL3: nil
1 gal jug UL4: nil
1 gal jug SK0: nil
1 gal jug SK1: nil
- Extraction Jar: Fenugreek/Vodka extraction (2011-12-15)
Planning
Add your name to the bottom to secure a spot in the brew queue. If you need to insert a priority batch, post to the mailing list & we'll hash it out.
ClintonEbadi Pumpkin Brown Ale
SteveKillen President's Honey Ale 10 gallons
RachelBrown Maple Nut Brown Ale
- Luke's Ghost Pepper Porter
Competing
Piedmont Brewer's Cup, 2011-10-22
SteveKillen's Bubonic Porter #1 PBC-Bubonic.pdf Score: 30 (Scoresheets: PBC-porter-1.png PBC-porter-2.png)
SteveKillen's Luna Moth #2 PBC-LunaMoth.pdf Score: 32 (Scoresheets: PBC-saison-1.png PBC-saison-2.png)
Natty Greene's Monthly Homebrew Contest, 2012-6-26
ClintonEbadi's Nowruz [w/Belgian yeast]
BtTempleton's Biere de Garde
SteveKillen's Irish Red [w/Scottish yeast] 3rd place (tied) in the popular vote!
SteveKillen's Stovetop Saison
NC State Fair, Oct 2012
SteveKillen Persephone RIS
RachelBrown Luna Moth Saison
Piedmont Brewer's Cup, 2012-10-20
SteveKillen's Bubonic Porter (Wyeast 1028) Score: 29
SteveKillen's Peach Pale Ale Score: 33
Useful Information
Required Reading
Read How to Brew. Do not continue, do not pass Go until you've at least made it through Chapter One.
Styles/Ingredients
Hopunion Variety Databook has tons of useful info on different hop varieties1
The Hop FAQ, revised by Glenn Tinseth himself. Hop wizardry inside!
General Brewing
Mr Malty has a few useful articles
A guide to off flavors in beer
Technique
A useful guide to late-hopping/hopbursting
A thread on HomebrewTalk about Stovetop All-grain batches
A handy chart to help you determine your Hop Schedule courtesy of http://www.brewsupplies.com/
A guide to calculate the sugar contribution of various fruits
Software
Brewtarget A newer Free Software brewing tool. Looks similar to QBrew, but apparently has no shared codebase with it and is actively maintained.
QBrew recipe formulation tool and batch log. Available in Debian, and indispensable for the Free Software loving brewer. Lacking a few features compared to proprietary offerings, but bpt and ClintonEbadi are programmers... We don't use it anymore, but QBrew could be improved if one felt like it.
Online Calculators
Quick Infusion Calculator for temp adjustments with boiling water
Wine/Mead/Cider
Wine Links (has a lot of good stuff!)
The Bee's Lees, a Collection of Mead Recipes
The Home Winemaker's Manual. Not sure how popular, but looks to be the closest to How to Brew in terms of presentation of the process.
Location Specific
American Brewmaster Our friendly Local Home Brewing Store
Quality Brake and Inspection Center happens to fill up propane tanks (and for a good price: $17.50/20lbs as of 2010-11)
Water
All Grain Water Chemistry Somewhat applicable for extract brewing
Bru'N Water Knowledge Spreadsheet water calculator (it works with Libreoffice), but more importantly a much more thorough explanation of water chemistry than provided elsewhere or in howtobrew.
Cary and Raleigh use chloramines rather than chlorine for water purification. The homebrew shop was helpful and warned that this does not evolve out as gas during boiling like chlorine does, and results in band-aid like flavors in the final beer. To counteract this a quarter of a Campden tablet must be added for each five gallons of water used. This will react with the chloramines and cause them to evolve out as sulfur and chlorine gases within approximately fifteen minutes. The water may then be boiled as usual.
For extract brewing water adjustment is not essential, but water in the Raleigh region is soft enough that there will be issues with hop bitterness not being properly attenuated. The LHBS provided Water Crystals (8:1 CaSO4:MgSO4) with a recommended rate of one or two tsp per five gallons. A teaspoon has a mass of 4g (3.55...g:0.44...g).
Another thing to be wary of is steeping roasted grains in soft water:
Water chemistry also plays a role in tannin extraction. Steeping the heavily roasted malts in very soft water will produce conditions that are too acidic and harsh flavors will result. Likewise, steeping the lightest crystal malts in hard water could produce conditions that are too alkaline and tannin extraction would be a problem again. In this case, the terms Hard and Soft Water are being used to indicate a high (>200 ppm) or low(<50 ppm) level of carbonates and the degree of alkalinity of the brewing water.2
Raleigh
Important Water Ion Levels for Raleigh NC Water (mg/L)3 |
||||||
Date |
Ca+2 |
Mg+2 |
SO4-2 |
Na+ |
Cl- |
HCO3- |
2010 |
6.49 |
1.90 |
52 |
30.5 |
12.9 |
34.28 4 |
Jan 2012 |
8.5 |
4.0 |
47 |
34 |
15.9 |
43.55 4 |
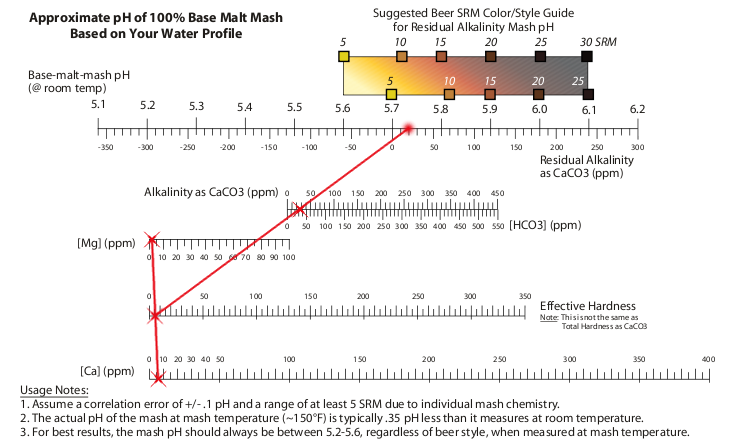
The water ion nomograph is pulled from Palmer's How To Brew. Looks like our water profile in Raleigh is good for pale, bitter beers; it's got a sulfate:chloride ratio of 3:1 (maybe -- it also has very little of either). To make darker, maltier beers, some additions will be required. SteveKillen estimates that for our 8ish-gallon water volume, a total of 3.6g chalk, 3.6g baking soda, and 4g of non-iodized salt will provide a better water profile for malty beers like the Bubonic Porter.3 Further tweaking may be required, but a good goal is to test several styles of beer and come up with a handful of good generic numbers.
Cary
Important Water Ion Levels for Cary NC Water (mg/L)3 |
|||||
Ca+2 |
Mg+2 |
SO4-2 |
Na+ |
Cl- |
HCO3- |
9.60 |
2.90 |
38 |
32.1 |
21.3 |
50.4 4 |
With 2g of Water Crystals |
|||||
25 |
4 |
78 |
32 |
21 |
50 |
With 4g of Water Crystals |
|||||
41 |
5 |
119 |
32 |
21 |
50 |
With 8g of Water Crystals |
|||||
72 |
6 |
201 |
32 |
21 |
50 |
Sulfate:Chloride ratio of raw water = (approximately) 1.2:1.0 which is not particularly good for anything. This should be closer to 2:1 for bitter ales, 1:3 for stouts and porters, and 1:2 for milder ales (e.g. Justin's Brown Ale). Some minor adjustment with NaCl will be needed; since extract is being used and the NaCl content is unknown a conservative approach is best. ClintonEbadi suspects aiming for a ratio of 2:1 for bitter beers, 1:2 for more malt dominated beers, and leaving it alone for the others is the best approach for now.
According to BeerSmith our Magnesium levels are insufficient for yeast nutrition, and the sulfate levels are too low for bitter beers (like ClintonEbadi's Pale Ale recipe).
Based upon this information it appears that a good approach will be to add 2g of Water Crystals to all batches with an appropriate amount of NaCl to balance the additional Sulfate.
Future Ideas
General Beer
Beer Advocate a number of reviews include a description of the hop varieties and grains used--this may be helpful!
Available Equipment
Most owned by ClintonEbadi; all available for shared use.
Wort Production
Bayou Classic KAB5 Low Pressure Propane Burner. 100,000 BTU/h super burner. MOAR POWER
2 20lbs Propane Cylinders Just a basic propane cylinder
- places to get propane refill: UHaul on Capital (north of 440), BJs on Old Wake Forest off Capital (south of 540)
2'x2' Paving Stone to provide a stable surface for the burner and protection for deck wood against drying out or scorching
15 gallon Mash Lautering Tun with fancy copper manifold for high-gravity brewing (OG > 1.060) & 10-gallon batches.
- Total materials cost ~$95
- 60qt cooler = $50
- Initial parts bill $56
- 1 unused 10' copper tube -$12
- misc. fenders from Ace
5 Gallon Mash Lautering Tun with stainless steel braid for masochism and all-grain batches (OG <= 1.060)
- Still useful for lower-gravity batches; better overall efficiency and less head space = great heat retention!
Parts list, total cost ~$40.
Make sure to check out a better guide to the actual construction.
Stainless steel 15.5 gallon keggle courtesy of Aviator Brewing and Steve's dremel!
Polarware 30 quart stainless steel brew kettle. Basic, but workable for full five gallon wort boils. LHBS recommended the low end kettle and reserving funds for a future purchase of a fancy ten gallon kettle.
Immersion Wort Chiller. Just a basic coiled copper tubing gadget.
Northern Tool Submersible Pump For circulating ice water through the immersion chiller in an attempt to save water. Cost about the same as the pond pump that didn't work, but a quick test was promising--the flow rate through the wort chiller looked as high as with the garden faucet. As of 2011-06-09 we have successfully cooled around 7 brews with the pump and it has yet to break--we can cool a five gallon batch to 74F in ~15 minutes using only 20 gallons of water and eight frozen 2L soda bottles. If it doesn't break in six more monthsit's 2012-03-20 and the pump has survived a lot of batches so I think I can recommend it.
48" Stirring Spoon That's what she said
Kettle Hop Bag Holder For containing the massive amount of hops in a ten gallon batch. Worked pretty well for /Recipes#Nowruz, but the hops themselves still absorb quite a bit of water on their own.
Fermentation
Ranco Digital Temperature Controller for the fridge at Evergreen (Steve's)
6 6 Gallon Better Bottles (2 Steve's, 2 Clinton's, 1 BPT's, 1 Sebastian's)
3 Gallon Better Bottle for experimental batches (Steve's)
7 Gallon Glass Carboy
5 Gallon Glass Carboy for secondary fermentation
5 1 Gallon Glass Jug for minibatches or starters
2 64oz Glass Jug (Rogue Dead Guy Ale Jug) for starters
Airlock enough for all fermentors and one starter jug (with stoppers for the carboys)
Siphoning &c
Autosiphon Fancy and new with a smaller outer tube diameter
Tee for splitting batches between fermenters (PROTIP: put the venturi tube above the tee)
VenturiTube gadget for wort aeration (2)
Faucet hose adapter. Easily acquired and infinitely useful -- the one we found has hose threads and a second set of smaller threads for an internally-threaded aerator. Really useful for filling buckets, using a jet bottle washer, getting a faster water flow for filling things (pasta pots, watering cans, ...).
Bucket Filler. Homemade hose end with siphon tubing attached to make filling five gallons of water a lot less of a pain in the ass.
Bottling
6 gallon bottling bucket with a spigot
Bottling Cane with an auto shutoff and whatnot
Bottle Capper basic wing capper
Colonna Capper/Corker Courtesy of BtTempleton
Drying Rack Plywood board with removable legs and holes for drying bottles after sanitization
Science!
Hydrometer for taking gravity readings
Floating Thermometer Warning: slow to read, not particularly accurate... mostly used to stir rehydrated yeast slurry now
12" SS Dial Thermometer Reasonably accurate, reads fast, can get deep into the mash
Pocket Kitchen Therometer Reasonable accurate, reads fast, handy for checking gravity sample temperatures
Cylinder not graduated, but with a 100mL (maybe--have to double check this) line marked. Used when taking gravity readings.
Wine Thief a small thief to take non-returnable samples for gravity readings
The Thief to take gravity readings in the fermenter!
Books
Beer Captured. A recipe book of 150 clone brews. Helpful as a basic guide for various styles.
Radical Brewing. A so far excellent source of ideas on weird (and not so weird) beer styles
&c
Sanitizer Bucket a.k.a five gallon plastic bucket from Home Despot
6 Gallon Ale Pail retired as a collection bucket for wort runnings from the mash tun
Things That Don't Actually Work
Igloo Lunchbox Cooler The top being uninsulated turned out to be a serious problem: the cooler lost around 40⁰F within 30 minutes. It turns out, however, that a metal pot in the 170⁰F oven maintained around 160⁰F and did not get too hot after all.
Smartpond 155GPH Submersible Pump for circulating ice water through the wort chiller. Low flow rate (not surprising), and broke after two uses. Garbage.
Obsoleted By the Ever-Forward March of Progress
Cooling system A pair of plant water drainage trays, tshirts, and a box fan... not as nice as a basement, but it actually works pretty well. (New place had a fridge so we grabbed a controller instead. This is still a great low-tech solution though--still in use by SteveKillen).
Expendables
That you can't eat at least.
5/16" ID Siphon Hosing
Bottlecaps about 24oz (6-7 batches worth), brightly colored (5/1/11)
NEEDED
More gadgets.
Like this one: Victoria Hand Mill, $30 after shipping. (out of stock)
Or this one: Another hand mill, $23.77 after shipping.
And this one: DIY Continuous Wort Hopper
Additions calculated using http://www.brewersfriend.com/water-chemistry/ which rounds to the nearest number... not so scientific, but good enough for our uses (3 4 5)